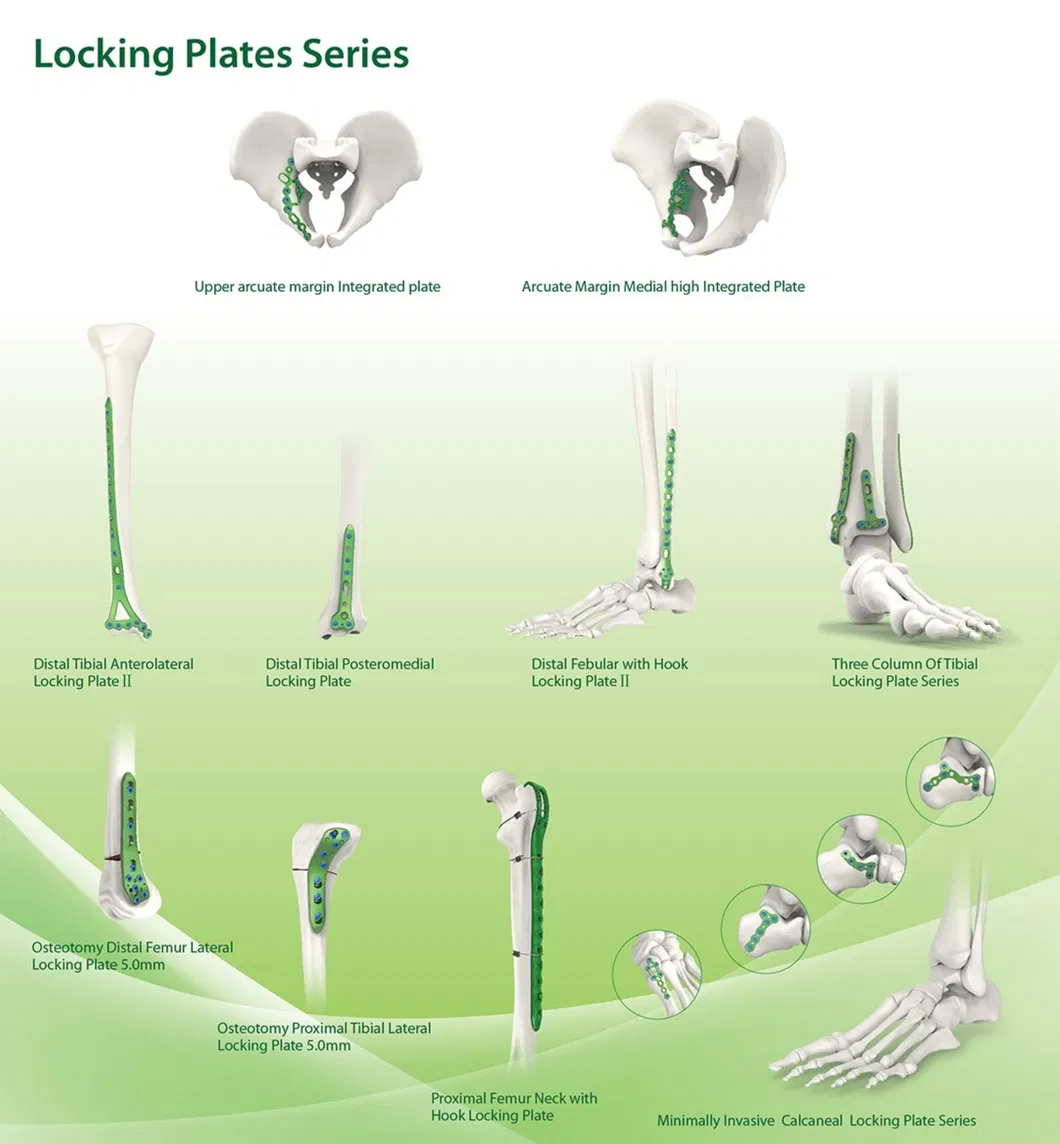
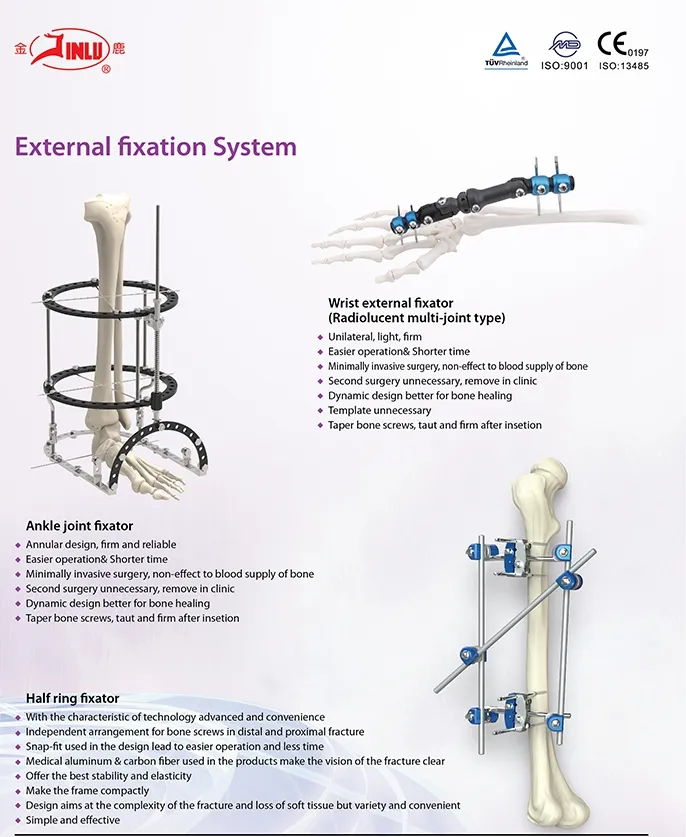
Established in 1958, the manufacturer specializes in the development, production, and distribution of orthopedic implants and specialized surgical instruments. Recognized as a key enterprise in the national medical apparatus sector, the company maintains a leading role within the industry. The primary product portfolio includes spinal fixation systems, interlocking intramedullary nails, trauma plates and screws, cannulated compression screws, external fixators, hip prostheses, and corresponding surgical tools.
Quality Assurance: All medical devices undergo rigorous testing and meet national and provincial quality standards. Products feature laser-etched material codes and anti-counterfeit marks to ensure traceability and reliability.
The manufacturing facility is equipped with advanced imported CNC machinery, high-precision digital control systems, and milling equipment. Production occurs within a Grade 100,000 cleanroom purification environment to guarantee product sterility and safety. As an ISO 9001 certified entity, the group has received multiple awards for scientific and technological innovation, reflecting a commitment to superior design and engineering excellence.
❓ Frequently Asked Questions
1 What types of orthopedic products are available?
The catalog includes metal bone plate systems, cannulated bore screws, intramedullary nails, spinal fixation devices, and external fixation tools.
2 How is product quality guaranteed?
All products are manufactured according to CE standards. We conduct pre-production sample reviews and a final inspection of every batch before shipment.
3 Are OEM and customization services accepted?
Yes, we provide full OEM and customization services to meet specific clinical requirements and brand specifications.
4 What materials are used for the implants?
Our implants are primarily crafted from high-grade Titanium Alloy and Stainless Steel to ensure biocompatibility and structural integrity.
5 What international standards do the products meet?
The factory and products are certified under ISO 9001, ISO 13485, and CE, ensuring compliance with global medical device regulations.
6 What is the standard packaging for shipping?
Items are securely packed in protective plastic bags and further reinforced with standard export cartons to prevent damage during transit.