Who are we?
We are a manufacture of orthopedic implants and instruments since 1958. Our main products include trauma plates, interlocking nails, spinal fixators, external fixators and power tools.
How can we guarantee quality?
Our products are produced according to EU CE standards. We always provide a pre-production sample before mass production and conduct a final Inspection before shipment.
What can you buy from us?
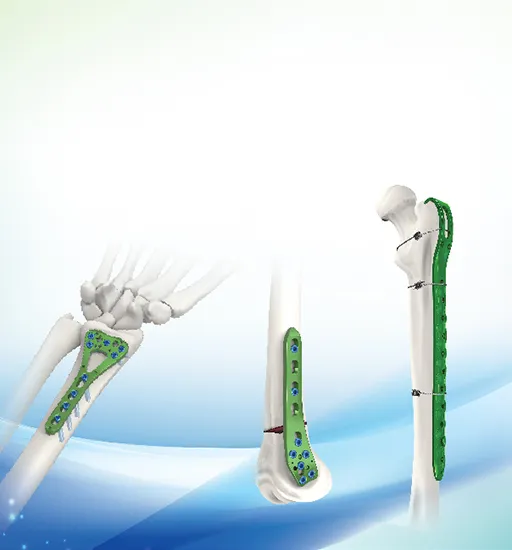
You can purchase Metal Bone Plates Screw Systems, Metallic Cannulated Bore Screws, Metallic Interlocking Intramedullary Nails, Spinal Fixation Devices, and External Fixation Devices with Needles.
Why should you buy from us not from other suppliers?
With decades of experience since 1958 in R&D and manufacture of orthopedic implants, we maintain a strict quality control system with CE/ISO/GMP certificates and excellent service.
What services can we provide?
Accepted Delivery Terms: FOB, CIF, EXW, Express Delivery; Accepted Payment Currency: USD, EUR, CNY; Accepted Payment Type: T/T, L/C, Western Union, Cash, Escrow.
Which languages are spoken for communication?
Our team can provide services and support in English and Chinese.