


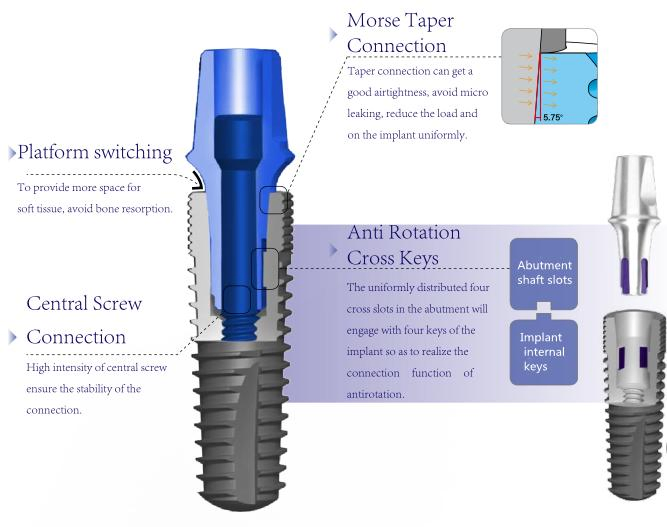
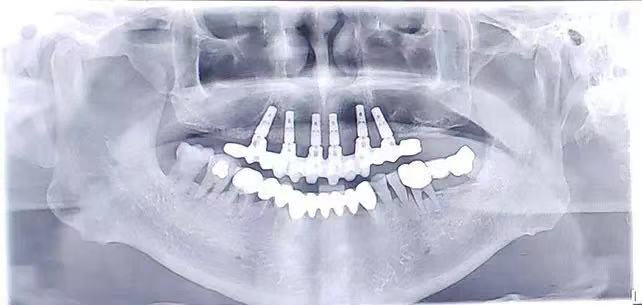










| Material | IV Pure Titanium |
| Applicable Departments | Dental Department |
| Nature | Implant Materials & Artificial Organs |
| Certification | CE, FDA |
| Properties | Implant Materials & Artificial Organs |
| Type | Implantable Artificial Organs, Submerged Type |
| Instrument Classification | Class III |
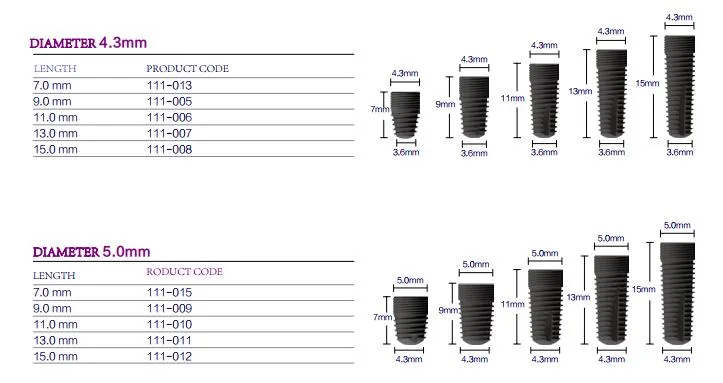
| Diameter | 3.4mm / 3.8mm / 4.3mm / 5.0mm |
| Length | 9mm / 11mm / 13mm / 15mm |
| Feature | Reverse Taper / Straight / Taper Design |
| Single Gross Weight | 0.020 Kg |
| Transport Package | Wooden Box Suitable for Long-Distance Transportation |
| HS Code | 9021100000 |
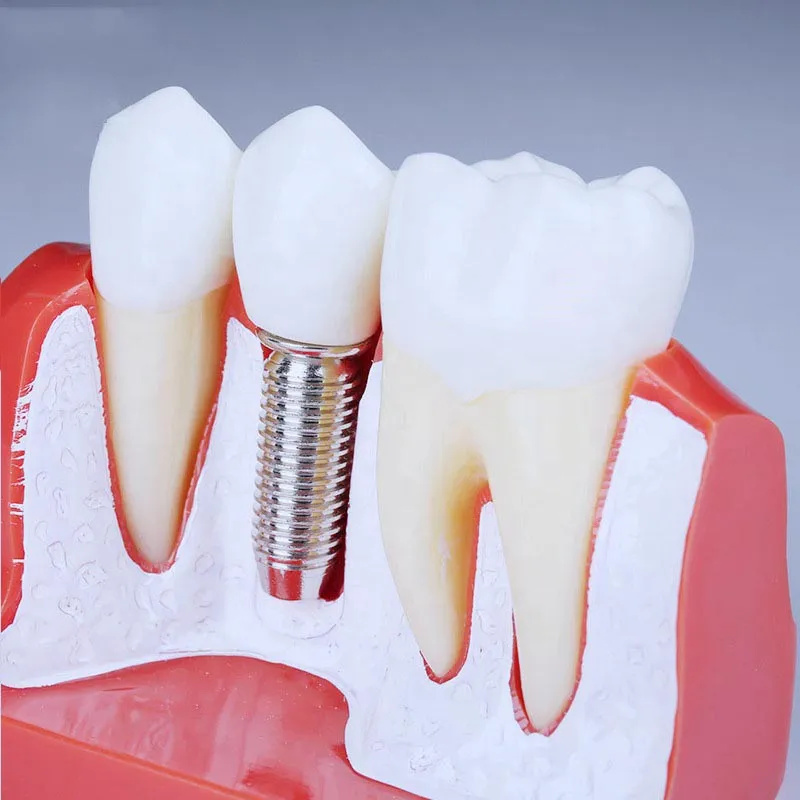


Our implants use premium Pure Titanium IV, ensuring the highest level of biocompatibility and strength for long-term dental restoration.

Advanced manufacturing equipment and a proven quality assurance system produce world-class mainstream dental implant systems. All implants are processed in an entire seal asepsis production workshop.

Established in 2005, the facility represents a significant investment in surgical needles and sutures. The main product line covers surgical sutures, surgical needles, and advanced dressings, aiming to be a powerful manufacturing base in the medical field.



Our products are certified by ISO 13485 Quality Management systems, ensuring all dental implants meet rigorous international standards for safety and efficacy.
