














| Products | Description |
|---|---|
| Cementless Femoral Stem | 8# (Φ5.7 × 115 × 135°) |
| 9# (Φ5.1 × 130 × 135°) | |
| 10# (Φ5.5 × 140 × 135°) | |
| 11# (Φ6.5 × 145 × 135°) | |
| 12# (Φ7.5 × 150 × 135°) | |
| 13# (Φ8.4 × 155 × 135°) | |
| 14# (Φ9.1 × 160 × 135°) | |
| 15# (Φ10.0 × 165 × 135°) | |
| 16# (Φ11.2 × 170 × 135°) |
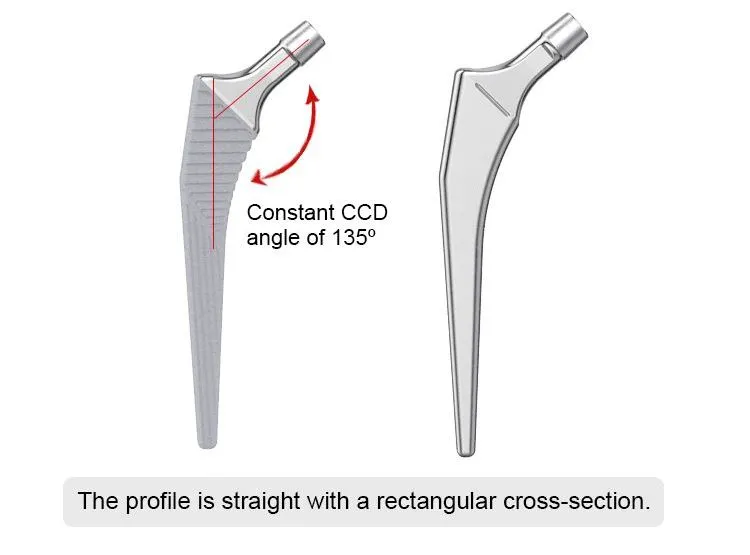
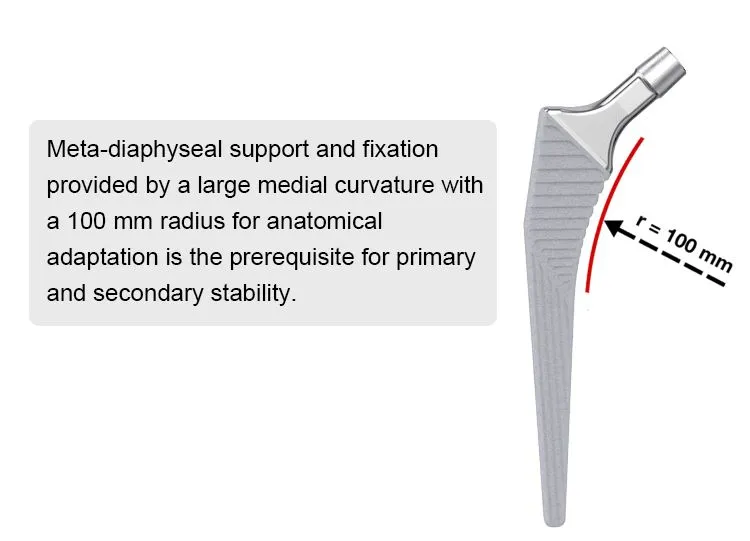

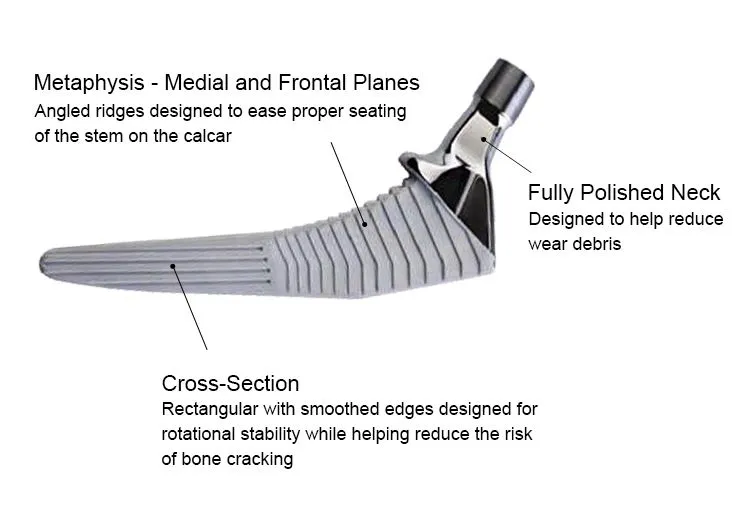
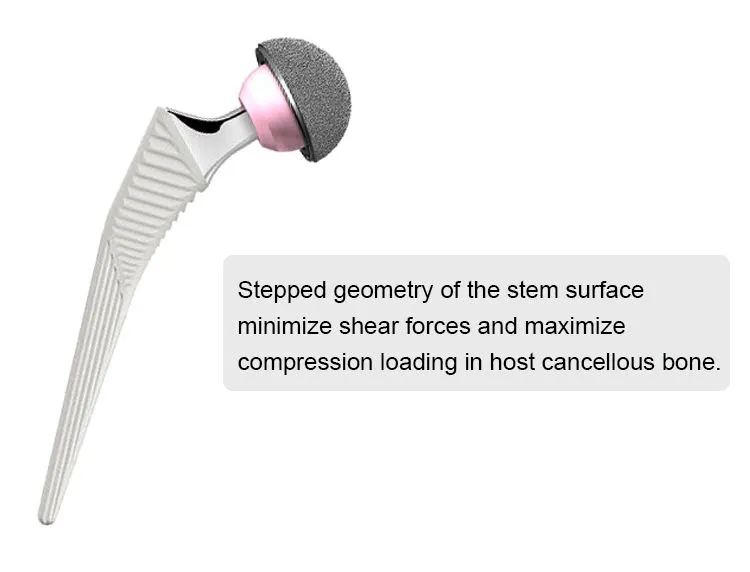
This cementless femoral stem is designed for total hip arthroplasty, providing stable primary fixation and promoting long-term biological fixation through bone ingrowth. Made from high-grade titanium alloy (Ti6Al4V), it offers excellent strength, corrosion resistance, and biocompatibility. The anatomical shape and porous coating ensure optimal fit and osseointegration. Suitable for primary hip replacement in patients with good bone quality.
Serving 2,500+ clients across 70+ countries.

CE & ISO Approved

Cutting Edge Equipment
1. Comprehensive Warranty: Full one-year warranty with reliable maintenance services to keep your product performing at its best.
2. Customer Engagement: Regular feedback collection to continuously optimize products based on real surgical experiences.
3. Industry Expertise: Professional training courses and conferences to stay updated with latest developments.
On-time Delivery, Safe Choice

As a leader in orthopedic implants and instruments manufacturing, we have successfully supplied high-quality solutions for over 14 years. With cutting-edge equipment and sales offices established in major medical hubs, we offer products of the highest industrial standards through a mature supplier system. We are continually pushing the limits of our know-how to provide innovative solutions worldwide.

Exhibiting for 10 Consecutive Years
